The Mother Lode of Mutations
Survey of Zebrafish Early Development Potentially
Relevant for Understanding Human Infertility and Birth
Defects
(Philadelphia, PA) -- After five years and thousands
of zebrafish breeding experiments, Mary C. Mullins,
PhD, associate professor of cell and developmental
biology at the University of Pennsylvania School
of Medicine and colleagues have published a
description of dozens of mutations that will help determine
the earliest steps in vertebrate development, which
take the spherical embryo to a complex creature. These
discoveries are described in a pair of papers in the
June Developmental Cell and are featured on
the cover of that issue. In time, these discoveries
may help researchers understand human sterility and
fertility problems, as well as what causes certain birth
defects.
Molecular control of the step-by-step process of how
the zebrafish body unfolds relies to some extent on
maternally driven processes. These depend on proteins
derived only from the egg, which are critical to embryonic
development prior to the activation of the new embryo’s
own genome. In vertebrates, maternal gene products direct
such essential processes as fertilization; the first
cellular divisions of the embryo; and the head-to-tail
arrangement of developing cells. The genes also control
morphogenetic movement, the migration of cells to form
the three-dimensional structure of the embryo.
In 1998 the Mullins lab embarked on a large-scale maternal-effect
mutant screen, not previously performed in a vertebrate
animal model. In addition to the 68 maternal mutations,
through inbreeding studies, they also discovered five
paternally derived mutations. Daniel S. Wagner,
PhD, and Roland Dosch, PhD,
both postdoctoral fellows in the Mullins lab, spent
five-plus years each on this intensive research project.
“These maternal processes are well-studied in
invertebrates, but not vertebrates,” says Mullins.
“Genetic screens have been extremely powerful
in identifying key genes and understanding the processes
involved.” This collection of mutants provides
the first molecular analysis of maternal control of
embryonic development in vertebrates.
The process is time-consuming, yet has been worth the
wait. Mullins uses a process in which genes are mutated
at random with a chemical mutagen to determine what
genes are required for a particular process of interest.
This is opposed to reverse genetics, where there’s
a particular gene of interest, which is then knocked
out to determine its function. The mother—who
is the mutant—is bred with a male non-mutant.
“All of her progeny are affected because the father
doesn’t provide any mutant genes,” explains
Mullins. “It’s the mother who’s providing
what’s in the egg.” The Mullins group can
ultimately identify the genes because the almost-completed
zebrafish genome sequence is available in public databases.
The first paper describes 21 maternal-effect mutants
from the earliest stages of embryonic development, when
it is literally only hours old. These mutants –
some dubbed over easy, soufflé, sunny side
up, jumpstart, and buckyball – include
such basic processes as the first embryonic cellular
divisions and the initial head-to-tail arrangement of
cells.
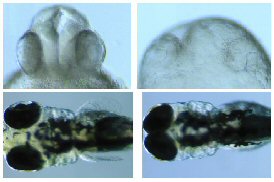 In
addition to mutants that affect basic patterning, or
assembly, of embryonic tissue the researchers describe
in the second paper some “totally unexpected”
mutants from one-day-old embryos, explains Mullins.One
was a morphogenesis mutant called pollywog.
(Top; click on thumbnail image to view full-size photos).
This gene is involved in orchestrating the ultimately
three-dimensional character of the head. “We end
up with flat-headed fish,” says Mullins. “The
head is just spread out on the yolk and it doesn’t
elevate to look like the normal three-dimensional structure.”
The group surmises that this is a cell movement defect.
Another mutant, dubbed pug, affected the embryonic
body plan. (Bottom images). Embryos from pug
mutant mothers have no pectoral fins – the first
limbs of zebrafish that form – and narrow-set
eyes, as well as defects in the midbrain and cerebellum.
In
addition to mutants that affect basic patterning, or
assembly, of embryonic tissue the researchers describe
in the second paper some “totally unexpected”
mutants from one-day-old embryos, explains Mullins.One
was a morphogenesis mutant called pollywog.
(Top; click on thumbnail image to view full-size photos).
This gene is involved in orchestrating the ultimately
three-dimensional character of the head. “We end
up with flat-headed fish,” says Mullins. “The
head is just spread out on the yolk and it doesn’t
elevate to look like the normal three-dimensional structure.”
The group surmises that this is a cell movement defect.
Another mutant, dubbed pug, affected the embryonic
body plan. (Bottom images). Embryos from pug
mutant mothers have no pectoral fins – the first
limbs of zebrafish that form – and narrow-set
eyes, as well as defects in the midbrain and cerebellum.
Because the process that Mullins used to obtain her
mutants involved three generations before she could
test for a mutant mother, she didn’t know how
frequently such mutant moms would be produced or be
able to breed. “That was the big risk of doing
the screen, a priori, you don’t know
what you’re going to get,” says Mullins.
“Now we feel we have this little gold mine.”
Penn researchers Keith A. Mintzer, Greg Runke, and Anthony
P. Wiemelt were also co-authors on the papers. This
work was funded in part by the National Institutes of
Health, the March of Dimes Birth Defects Foundation,
and the American Cancer Society.
For
a printer friendly version of this release, click
here.
###
PENN Medicine is a $2.5 billion
enterprise dedicated to the related missions of medical
education, biomedical research, and high-quality patient
care. PENN Medicine consists of the University of Pennsylvania
School of Medicine (founded in 1765 as the nation’s
first medical school) and the University of Pennsylvania
Health System (created in 1993 as the nation’s
first integrated academic health system).
Penn’s School of Medicine is ranked #3 in the
nation for receipt of NIH research funds; and ranked
#4 in the nation in U.S. News & World Report’s
most recent ranking of top research-oriented medical
schools. Supporting 1,400 fulltime faculty and 700 students,
the School of Medicine is recognized worldwide for its
superior education and training of the next generation
of physician-scientists and leaders of academic medicine.
Penn Health System consists of four hospitals (including
its flagship Hospital of the University of Pennsylvania,
consistently rated one of the nation’s “Honor
Roll” hospitals by U.S. News & World Report),
a faculty practice plan, a primary-care provider network,
three multispecialty satellite facilities, and home
health care and hospice.
Penn Medicine is one of the world’s leading academic medical centers, dedicated to the related missions of medical education, biomedical research, excellence in patient care, and community service. The organization consists of the University of Pennsylvania Health System and Penn’s Raymond and Ruth Perelman School of Medicine, founded in 1765 as the nation’s first medical school.
The Perelman School of Medicine is consistently among the nation's top recipients of funding from the National Institutes of Health, with $550 million awarded in the 2022 fiscal year. Home to a proud history of “firsts” in medicine, Penn Medicine teams have pioneered discoveries and innovations that have shaped modern medicine, including recent breakthroughs such as CAR T cell therapy for cancer and the mRNA technology used in COVID-19 vaccines.
The University of Pennsylvania Health System’s patient care facilities stretch from the Susquehanna River in Pennsylvania to the New Jersey shore. These include the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, Chester County Hospital, Lancaster General Health, Penn Medicine Princeton Health, and Pennsylvania Hospital—the nation’s first hospital, founded in 1751. Additional facilities and enterprises include Good Shepherd Penn Partners, Penn Medicine at Home, Lancaster Behavioral Health Hospital, and Princeton House Behavioral Health, among others.
Penn Medicine is an $11.1 billion enterprise powered by more than 49,000 talented faculty and staff.